The misuse of antibiotics is the single greatest driver of bacterial resistance worldwide. Understanding the biological mechanisms through which bacteria develop resistance helps explain why responsible antibiotic use is so critical. This is not complicated science reserved for laboratories. It is a process with direct implications for every patient in Nigeria and beyond.
The Fundamentals of Antibiotic Resistance Mechanisms
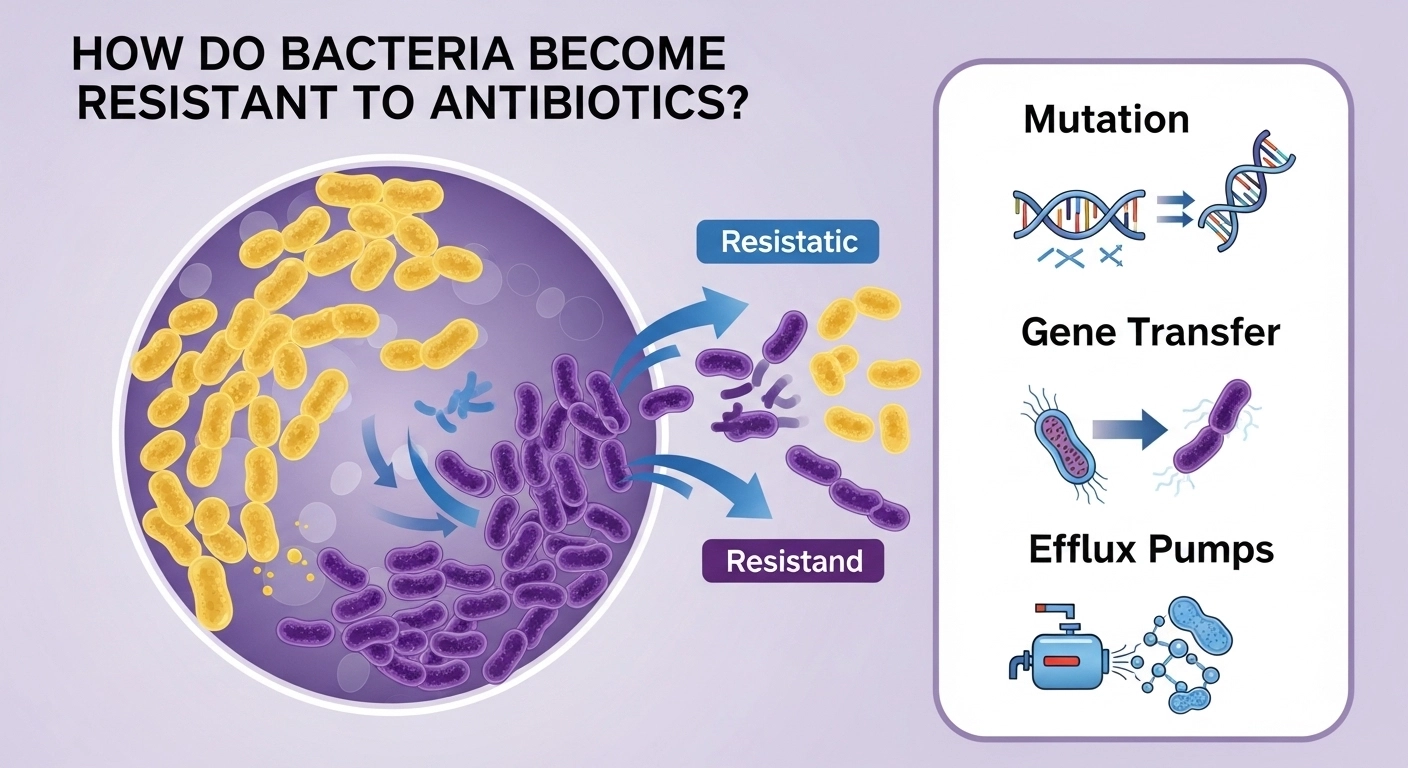
How Bacteria Become Resistant: Mutation
Bacteria reproduce at an extraordinary speed. A single bacterium can produce millions of offspring within hours. During replication, random genetic mutations occur. Most mutations are harmful to bacteria, but some happen to reduce the bacterium’s sensitivity to antibiotics.
When antibiotics are present, susceptible bacteria die while those with resistance mutations survive and reproduce. This is natural selection operating at a microbial scale, and it happens every time antibiotics are used inappropriately.
Horizontal Gene Transfer: Sharing Resistance
Bacteria do not only inherit resistance from parent cells. They can also acquire resistance genes from neighbouring bacteria through a process called horizontal gene transfer. This means one resistant bacterium can spread its resistance traits to many others very rapidly.
This mechanism explains why resistance can appear in a bacterial population even before antibiotic exposure. The resistance genes may already exist in the environment, waiting to be transferred.
Key Antibiotic Resistance Mechanisms Explained
Enzymatic Inactivation
Some bacteria produce enzymes that chemically break down antibiotics before they can do any damage. Beta-lactamase enzymes, for example, destroy penicillin-based drugs. Extended-spectrum beta-lactamases (ESBLs) can neutralise a wide range of antibiotics simultaneously.
ESBL-producing bacteria are increasingly detected in Nigerian hospitals and are classified as a major public health threat because they render many standard antibiotics useless.
Target Modification
Antibiotics work by binding to specific bacterial structures. Some bacteria alter those structures so the antibiotic can no longer attach effectively. Methicillin-resistant Staphylococcus aureus (MRSA) uses this mechanism to resist the entire class of penicillin-related antibiotics.
Efflux Pumps
Bacteria can develop internal pumps that actively expel antibiotic molecules before they reach their target. Efflux pumps effectively reduce the concentration of antibiotic inside the bacterial cell to a level that causes no harm.
These pumps are among the most adaptable resistance mechanisms because a single pump can expel multiple different types of antibiotics simultaneously.
Superbugs Explained: The Worst Outcome of Resistance
What Makes a Superbug?
A superbug is a bacterium that has developed resistance to multiple classes of antibiotics, often simultaneously. These multi-drug-resistant organisms are the most dangerous outcome of widespread antibiotic misuse.
Superbugs such as carbapenem-resistant Enterobacteriaceae (CRE) and drug-resistant Mycobacterium tuberculosis are already present in Nigeria. They represent a genuine therapeutic crisis because very few antibiotics remain effective against them.
Where Superbugs Emerge
Hospitals are primary breeding grounds for superbugs due to heavy antibiotic use and large concentrations of vulnerable patients. However, communities, farms, and water sources also harbour resistant bacteria.
NNAST works to monitor the spread of resistant organisms in Nigeria and supports policies to prevent their proliferation across healthcare and agricultural settings.
Why Understanding the Mechanism Matters for Prevention
Better Prescribing Saves Future Treatments
Each antibiotic belongs to a class. When resistance develops to one antibiotic within a class, it often affects others in the same group. Preserving antibiotic classes for future use requires strict discipline in how they are prescribed today.
Diagnostics Before Drugs
Laboratory-based diagnostics can identify the specific bacterium causing an infection and test which antibiotics it remains sensitive to. This approach, called culture and sensitivity testing, is the gold standard for preventing unnecessary antibiotic resistance and should be far more widely adopted in Nigerian healthcare settings.
Frequently Asked Questions
How quickly can bacteria become resistant to antibiotics?
Under the right conditions, bacteria can develop resistance within days. Research shows that detectable resistance can emerge in a bacterial population within as few as 11 days of antibiotic exposure when the drug is used incorrectly.
Are all superbugs untreatable?
Not entirely, but treatment options are severely limited. Some superbugs still respond to last-resort antibiotics such as colistin or linezolid. However, resistance to these drugs is also increasing, which makes the problem of antibiotic resistance increasingly urgent.
Can resistance genes survive outside a living bacterium?
Yes. Resistance genes can persist in soil, water, and organic matter for extended periods, even without a living host. This environmental reservoir of resistance genes poses a long-term public health challenge.